
Use the scrambler tool to randomly shuffle peptide or protein sequences.Ĭopyright © 2012-2023. Try the sequence splitter tool for fragmenting sequences of proteins and peptides with optional overlap. Peptide with Phosphoserine: Arg-Arg-Ala-Ser(PO3)-Pro-Val-Ala Also on this site 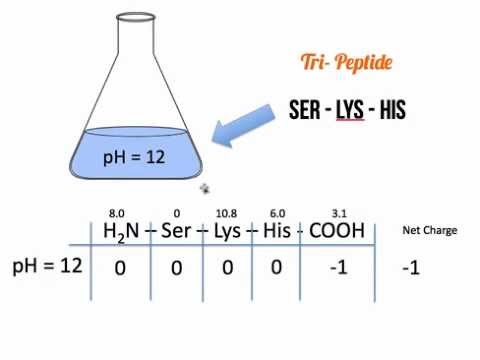
Peptide with isotopically labeled Valine and Isoleucine: looking up their pKa and calculating charge by henderson hasselbalch) and just add them up to get the total net charge. Peptide with disulfide bridge (1-6), capped at C-terminal (amide): Cyclic CYFQNCPRG-NH2 1 Answer Sorted by: 1 In theory, you can calculate the charge on every individual ionizable group (i.e. 3 Calculating pI valuesedit For an amino acidwith only one amineand one carboxylgroup, the pI can be calculated from the meanof the pKasof this molecule. But since most proteins are not only composed of one chain you cannot call them a peptide, but a polypeptide. Therefore if a protein were to contain only one strand of amino acids it could be called a peptide as you have noticed. Peptide with Biotin on N-terminal: Biotin-YGRKKRRQRRR When the environment is at a pH value equal to the proteins pI, the net charge is zero, and the protein is not bound to any exchanger, and therefore, can be eluted out. These proteins are also called polypeptides. About Relative Standard Deviation Calculator Basic AA: pI (pKa R group pKa Amino. Peptide with two disulfide bridges: ACDCRGDCFCG 2: Peptide bond forms between amino acid an antigen is a protein. A calculator would be used on the first term (to determine the value of 10 -15 ) while, by inspection, it is observed that the last three terms simply reduce to their respective. Head-to-tail cyclic peptide: cyclo(GRGDSP) peptide side-chain groups are those commonly reported in. The other two cases introduce other ionisable groups in the side chain ' R ' described by a third acid dissociation constant, pK a 3 acidic side chains The pI will be at a lower pH because the acidic side chain introduces an 'extra' negative charge. Linear peptide in three-letter code: Arg-Gly-Asp-Cys For the simplest amino acid, glycine, pKa1 2.34 and pK a 2 9.6, pI 5.97. Charge and isoelectric point of peptides are determined by the individual amino acids. Linear peptide in one-letter code: KYICNSSCM Peptide Charge and Isoelectric Point Shortcut. Please note: this is a rapidly changing project in development look, feel, and functionality may and will change. Take a look at the following posts for demonstration of what the calculator is capable of:įor a quick sample of the output screen, you can load a random peptide from the list of peptides the analytical tool has calculated before. The bond is a special linkage between the nitrogen atom of one amino acid and the carboxyl carbon atom of another. These chains contain two or more amino acids (forming amino acid polymers) that are coupled by a peptide bond. The peptide molecular weight calculator will display average and monoisotopic mass of the molecule, as well as a table of mass divided by charge values, both in positive and negative scan modes, which is useful for mass spec analysis. Both peptides and proteins fall under a category called polypeptide chains. 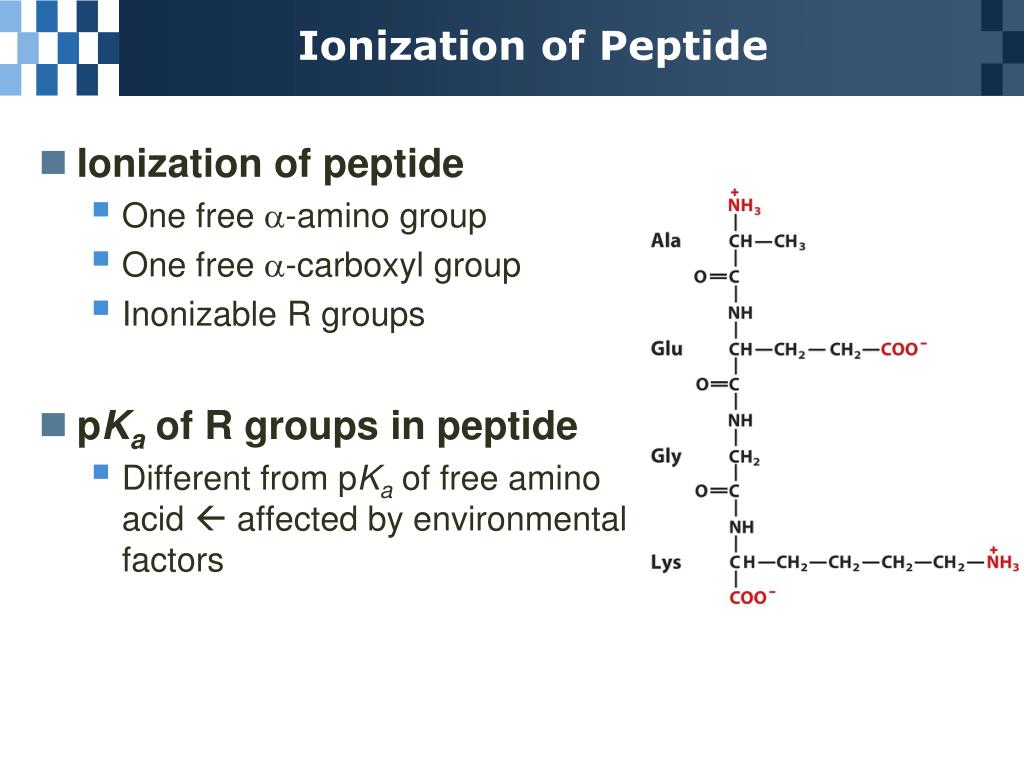
Simply type in, or copy and paste, peptide or protein fragment amino-acid sequence, including modifications, spacers, or special termini, and press the “Calculate” button. Approach 1 deprotonate e 3 (i.e. Let's call the ends e 1, e 2 and e 3 (from left to right). 2: Peptide bond forms between amino acid an antigen is a protein. According to orthocresol's answer: Since the p I is the p H at which the amino acid has no overall net charge, you need to average the p K a values relevant to the protonation/deprotonation of the form with no net charge. This procedure can of course be extended to the amino acids with acidic side chains (aspartic acid glutamic acid) and those with basic side chains (lysine arginine histidine).Sequence How to use this peptide analytical tool About Relative Standard Deviation Calculator Basic AA: pI (pKa R group pKa Amino. You'll find that since the side chain has a lower $\mathrm pK_\mathrm a$ than the amino group, you average the carboxyl and the side chain $\mathrm pK_\mathrm a$'s. The same logic applies to cysteine ( look up the $\mathrm pK_\mathrm a$ values and draw out the differently protonated forms). If the side chain $\mathrm pK_\mathrm a$ were lower than $9.11$, then you should average the carboxyl and side chain $\mathrm pK_\mathrm a$'s instead. It just so happens that $2.20$ is the carboxyl $\mathrm pK_\mathrm a$ and $9.11$ is the amino $\mathrm pK_\mathrm a$. Since the $\mathrm$ of tyrosine is $5.66$ (the average of $2.20$ and $9.11$).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
